All published articles of this journal are available on ScienceDirect.
Comparing Salivary Lactate Dehydrogenase Levels in Patients with Chronic Periodontitis with and without Atherosclerotic Cardiovascular Disease: A Cross-Sectional Study
Abstract
Background
Both chronic periodontitis and atherosclerotic cardiovascular disease are highly prevalent inflammatory diseases. The potential biomarker for these conditions is lactate dehydrogenase. The purpose of this study was to investigate salivary lactate dehydrogenase concentrations in patients with CP with and without ATH.
Methods
This was a cross-sectional study involving 87 participants aged 30-60 years, divided into four groups: the ATH-only group (n=25), the CP-only group (n=25), the ATH+CP group (n=25), and the normal control group (n=12). The participants underwent periodontal examination and sampling of unstimulated saliva. Salivary LDH was analyzed by ELISA. Statistical analysis included one-way ANOVA, post hoc tests, Pearson correlation, and ROC curve analysis.
Results
The salivary LDH values were significantly different among the groups (p < 0.001). The ATH+CP group had the highest value of salivary LDH (24.55 ± 2.55 U/L), followed by the CP group (13.44 ± 4.27 U/L), the ATH group (11.68 ± 3.86 U/L), and the control group (1.53 ± 0.31 U/L). The values of salivary LDH were positively correlated with the clinical parameters of periodontal disease in the diseased groups. The diagnostic accuracy of the ROC analysis was excellent, with an AUC = 0.987.
Discussion
The results obtained in this study suggest that salivary LDH is potentially useful as a noninvasive biomarker for estimating inflammation in patients with periodontitis and cardiovascular disease. The increased levels of LDH, particularly among patients with combined conditions, suggest pathophysiological connections and support emerging evidence concerning the bidirectionality of associations between oral and systemic health.
Conclusion
Salivary LDH is a potential noninvasive biomarker of periodontal disease and may reflect the inflammatory burden in patients with concomitant ATH. The presence of elevated LDH in patients with both conditions indicates pathophysiological overlap.
1. INTRODUCTION
Periodontal disease affects millions of people around the world and is one of the most common chronic inflammatory conditions in humans. Among adults, chronic periodontitis is the leading cause of tooth loss [1, 2]. The disease develops when anaerobic gram-negative bacteria colonize the dental biofilm, triggering a chronic infection that gradually destroys the structures supporting the teeth. Patients typically present with alveolar bone loss and deepened periodontal pockets [3, 4]. Although bacteria initiate the disease process, their progression and severity depend heavily on the host's immune response, along with environmental and behavioral factors [5].
Cardiovascular disease continues to be the leading cause of death worldwide. Atherosclerotic cardiovascular disease alone affects approximately 17.3 million people each year, accounting for nearly one-third of all deaths globally [6]. Experts predict that this number will increase to 23.3 million by 2030 as populations age and rates of obesity and metabolic disorders rise [7]. Coronary artery disease, which results from atherosclerosis, has multiple genetic contributors and remains a major cause of death in developing nations [8].
Chronic Periodontitis (CP) increases the risk of Atherosclerotic Cardiovascular Disease (ATH) through shared inflammatory pathways and the systemic circulation of periodontal pathogens and their products. This link is supported by evidence of periodontal pathogens in atherosclerotic plaques, increased blood inflammatory markers such as C-Reactive Protein (CRP), and impaired endothelial function. Managing periodontitis can improve cardiovascular risk markers, underscoring the importance of oral hygiene in preventing both conditions [1-4].
Lactate Dehydrogenase (LDH) is a cytoplasmic enzyme present in all human body cells. It is released into the extracellular environment following cell death and tissue damage, where it can be detected in various body fluids, including saliva and gingival crevicular fluid. In the context of periodontal disease, elevated salivary LDH levels indicate periodontal tissue destruction. In parallel, serum LDH is a well-established biomarker for myocardial injury and is used in the diagnosis and prognosis of patients with coronary atherosclerosis. Given its role as a marker of cellular damage in both conditions, salivary LDH is a compelling noninvasive biomarker for assessing the cumulative inflammatory burden in patients with both CP and ATH [9].
In the present century, inflammatory reactions play a key role in the pathogenesis of diseases, especially those involved in the etiology and progression of periodontal and cardiovascular diseases [10]. The immunology of atherosclerosis comprises both the innate and adaptive immune systems, and this response is central to how the disease develops and progresses clinically, i.e., heart attack [11]. A variety of immune cells, macrophages, and T and B cells, are involved in this process, which leads to the release of enzymes and cytokines that play a role in disease development [12].
Atherosclerosis arises when the endothelium, or inner lining of blood vessels, is impaired and inflammation becomes chronic. This leads to deposition of lipids within vessel walls, thereby forming atherosclerotic plaques [13, 14]. These plaques are morphologically characterized by a central lipid-rich core surrounded by foam cells (lipid-containing macrophages) capped with a fibrous cap [15]. Atherosclerosis exhibits inflammatory mechanisms highly similar to those seen in periodontal disease; the latter may therefore be mechanistically associated with the former [16].
Oral inflammation can exacerbate atherosclerosis by triggering both humoral and cellular immune responses [17]. Some of the proposed associations between periodontitis and atherosclerosis include the existence of shared risk factors, inflammation mediators, increased white blood cell counts and immune cell activation, and bacterial lipopolysaccharides from oral bacteria may be involved in systemic inflammation, as proposed by various studies conducted in this area [18].
The value of saliva as a diagnostic fluid has recently increased. It contains numerous compounds that may be used as biomarkers of systemic conditions, such as periodontal disease [19, 20]. The value of saliva lies in its ability to comprise materials from mucosal transudate and gingival crevicular fluid, making it a very effective fluid for analyzing pathogenic processes involved in various systemic conditions. Additionally, saliva collection is very affordable and painless, making it an excellent fluid for biomarker research [21, 22].
In recent years, numerous biomarkers have been identified for periodontal disease [23, 24]. Lactate dehydrogenase is one of the earliest biomarkers used as a diagnostic aid for periodontal disease [25]. This cytoplasmic enzyme is present in human body cells throughout all tissues. It is released into the extracellular environment following cell death and tissue damage, where it can be detected in saliva and gingival crevicular fluid [26, 27]. Consequently, LDH has been proposed as a diagnostic tool for assessing periodontal disease and evaluating the severity of myocardial injury in patients with coronary atherosclerosis [28].
Salivary LDH levels are crucial for determining the extent of periodontal tissue destruction. However, there remains a significant knowledge gap regarding salivary LDH levels in patients with chronic periodontitis who also have atherosclerotic cardiovascular disease. This study represents the first investigation of salivary LDH levels in Iraqi populations with these concurrent conditions. This research was limited by the absence of previous studies examining salivary LDH levels in atherosclerotic patients with chronic periodontitis and the critical importance of determining the degree of periodontal tissue deterioration in this population.
The null hypothesis for this study assumes that there is no significant difference in salivary LDH levels between patients with chronic periodontitis with and without atherosclerotic cardiovascular disease. This investigation aims to contribute to the growing body of evidence supporting the bidirectional relationship between periodontal and cardiovascular diseases while exploring the potential utility of salivary LDH as a noninvasive biomarker for both conditions.
2. MATERIALS AND METHODS
2.1. Study Design and Ethical Considerations
This cross-sectional comparative study was conducted between July 2023 and October 2023, following approval from the Ethical Committee of the College of Medicine, University of Kufa (Reference number: MEC-48, Date: 26/6/2023). The Declaration of Helsinki and Good Clinical Practice guidelines were followed in this study. After receiving comprehensive information regarding the study objectives, procedures, potential risks, and benefits, all participants provided written informed consent.
2.2. Sample Size Calculation and Power Analysis
Sample size calculation was performed using G*Power (version 3.1.9.7) based on previous studies reporting large effect sizes (Cohen's d = 0.8) for LDH differences between periodontal disease patients and healthy controls [29]. With an alpha level of 0.05 and a power of 0.80, and accounting for a 10% dropout rate, the minimum required sample size was 20 participants per group. The actual sample sizes (25 participants per disease group, 12 per control group) exceeded these requirements, achieving a power of 0.95 to detect clinically meaningful differences.
2.3. Participant Selection and Study Groups
Eighty-seven participants aged 30–60 years were recruited for this study. Atherosclerotic cardiovascular disease patients were examined during catheterization procedures at the Al-Sadar Teaching Hospital Heart Center, Iraq. Chronic periodontitis patients and healthy subjects were recruited from the Periodontology Department at the Faculty of Dentistry, University of Kufa, Iraq (Fig. 1).
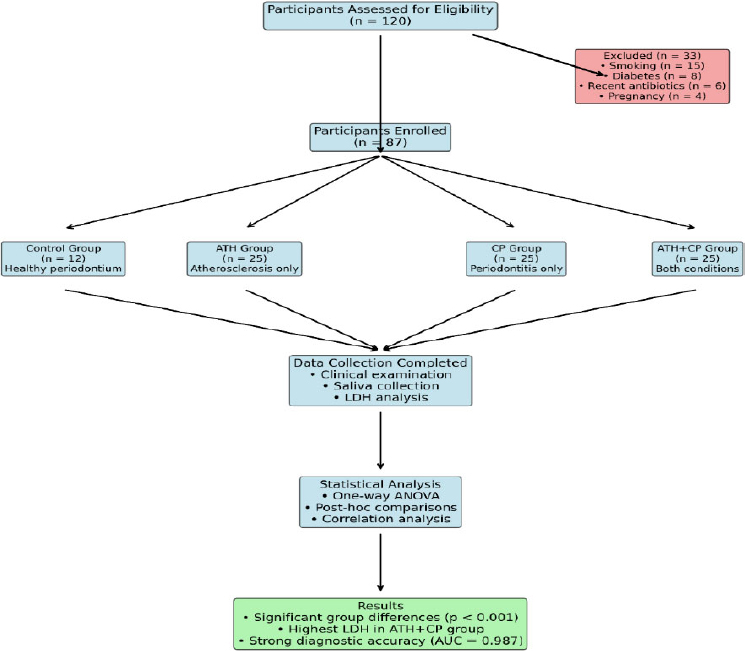
Study participant flow chart showing recruitment, exclusion criteria, group allocation, and analysis procedures.
The participants were allocated into four distinct groups based on their medical and periodontal status:
2.3.1. Group I (ATH, n=25)
Individuals with confirmed atherosclerotic cardiovascular disease who had undergone cardiac catheterization within the previous year, were receiving anticoagulant medication (Plavix), and demonstrated at least one coronary artery with ≥50% stenosis while maintaining a clinically healthy periodontium.
2.3.2. Group II (CP, n=25)
Individuals with chronic periodontitis without systemic diseases. In accordance with the Lang et al. criteria [30], participants in this group presented with more than four periodontal pockets >4 mm deep, associated with clinical attachment loss (CAL) of 1–2 mm or greater.
2.3.3. Group III (ATH+CP, n=25)
Individuals who presented with chronic periodontitis and atherosclerotic cardiovascular disease and met the criteria for both conditions described above.
2.3.4. Group IV (Control, n=12)
Healthy individuals serving as controls, with Gingival Index (GI) values <0.5 indicating clinically healthy periodontia [31]. The control participants met two additional requirements: absence of periodontal pockets and no history of systemic disorders.
2.4. Inclusion and Exclusion Criteria
2.4.1. Inclusion Criteria
The inclusion criteria were as follows:
- Age between 30–60 years
- Ability to provide informed consent
- Willingness to participate in all study procedures
- For ATH groups: confirmed atherosclerotic cardiovascular disease with catheterization evidence
- Clinical and radiographic evidence of chronic periodontitis in the CP group
2.4.2. Exclusion Criteria
The exclusion criteria were as follows:
- History of systemic diseases, including rheumatoid arthritis, diabetes mellitus, autoimmune disorders, or other chronic inflammatory conditions
- Current smoking habit or smoking cessation within the previous six months
- Use of antibiotics or anti-inflammatory medications within the preceding three months
- Periodontal treatment received within the previous three months
- Pregnancy or lactation
- Use of oral contraceptive medications
- Presence of acute periodontal conditions or oral infections
- Inability to provide adequate saliva samples
2.5. Clinical Examination and Data Collection
A specially designed case report form was completed for each participant, documenting demographic information, comprehensive medical and dental history, current medications, and relevant clinical findings. All clinical examinations were performed by calibrated examiners who received training and calibration exercises to ensure measurement reliability and consistency.
The periodontal status assessment included a comprehensive evaluation using the following clinical parameters:
2.5.1. Plaque Index (PLI)
PLI was assessed via O'Leary's method (1972) with disclosing agent application [32]. The presence or absence of plaque was recorded at four surfaces of each tooth (buccal, palatal/lingual, mesial, and distal), and the percentage of plaque-positive surfaces was calculated.
2.5.2. Gingival Index (GI)
Evaluated according to the Löe and Silness criteria [33], gingival inflammation was assessed on a scale from 0 (no inflammation) to 3 (severe inflammation) at four sites per tooth.
2.5.3. Pocket Depth (PPD)
PPD was measured via a Williams periodontal probe (marked at 1, 2, 3, 5, 7, 8, 9, and 10 mm) from the gingival margin to the base of the periodontal pocket. Six measurements per tooth were recorded (mesiobuccal, buccal, distobuccal, mesiolingual, lingual, and distolingual).
2.6. Saliva Collection and Processing
Unstimulated whole saliva samples were collected from all participants following standardized protocols to ensure sample quality and consistency. The participants were instructed to avoid eating, drinking, smoking, or performing oral hygiene procedures for at least two hours before sample collection. The collection was carried out between 9:00 AM and 11:00 AM to minimize circadian rhythm effects on salivary composition.
The participants were seated comfortably and instructed to allow saliva to accumulate in their mouths for two minutes before being expectorated into sterile collection tubes. A minimum volume of 2 mL was collected from each participant. Immediately after collection, the samples were placed on ice and transported to the laboratory within 30 minutes.
Saliva samples were centrifuged at 2000 rpm for 10 minutes at 4°C to remove cellular debris and mucins. The clear supernatant was carefully separated, aliquoted into sterile Eppendorf tubes, and then stored at -20°C until analysis. All samples were analyzed within one month of collection to ensure biomarker stability.
2.7. Lactate Dehydrogenase Analysis
Salivary LDH levels were quantified via a commercially available Enzyme-Linked Immunosorbent Assay (ELISA) kit designed for human LDH detection. The assay kits included reference standards, concentrated biotinylated detection antibodies, concentrated Horseradish Peroxidase (HRP) conjugates, sample diluents, HRP conjugate diluents, substrate reagents, concentrated wash buffers, stop solutions, plate sealers, and dismountable microplates.
The ELISA procedure was performed according to the manufacturer's instructions with the following modifications for optimal sensitivity:
2.7.1. Standard Preparation
Serial dilutions of the reference standard were performed in duplicate, with 100 μL applied to each well. Sample wells received 100 μL of diluted saliva samples.
2.7.2. Primary Incubation
Plates were sealed and incubated at 37°C for 90 minutes to allow antigen‒antibody binding.
2.7.3. Antibody Detection
After the liquid contents were removed, 100 μL of biotinylated detection antibody working solution was added to each well. The plates were sealed, mixed gently, and incubated at 37°C for 60 minutes.
2.7.4. Washing Steps
Wells were washed three times with 350 μL of wash buffer, with 1–2 minute soaking periods between washes. The residual liquid was removed using absorbent paper.
2.7.5. HRP Conjugate Incubation
100 μL of HRP conjugate working solution was added to each well, followed by sealing and incubation at 37°C for 30 minutes.
2.8. Quality Control and Assay Validation
The quality control measures included running duplicate samples, including positive and negative controls in each assay, and ensuring that all standard curves achieved correlation coefficients >0.99. Interassay and intra-assay coefficients of variation were maintained below 10% and 5%, respectively. The assay's detection limit was 0.1 U/L, with a linear range extending to 50 U/L.
2.9. Statistical Analysis
Statistical analysis was performed via SPSS software version 22.0. Data distribution normality was assessed via the Shapiro‒Wilk test. Primary analysis was performed via one-way Analysis of Variance (ANOVA) to compare LDH levels among the four study groups. The significance threshold was set at α = 0.05, and the chi-square test was used for categorical variables. Post hoc pairwise comparisons were performed via Tukey's honest significant difference test. Effect sizes were calculated via Cohen's d. Correlation analyses between LDH levels and clinical periodontal parameters were performed via Pearson correlation coefficients. ROC curve analysis was performed to evaluate the diagnostic accuracy of salivary LDH for the detection of periodontal disease. Statistical significance was set at p < 0.05.
3. RESULTS
3.1. Participant Characteristics and Baseline Demographics
The study successfully recruited 87 participants across four groups, with comprehensive baseline characteristics presented in Table 1. Significant age differences were observed among the groups (F = 28.261, p = 0.002), with the ATH+CP group being the oldest (57.33 ± 2.16 years), followed by the ATH group (50.53 ± 3.55 years), CP group (45.67 ± 3.14 years), and control group (32.17 ± 2.32 years). This age distribution reflects the natural progression and increased prevalence of both periodontal and cardiovascular diseases with increasing age.
| Characteristic | ATH (n=25) | CP (n=25) | ATH+CP (n=25) | Control (n=12) | p-value |
|---|---|---|---|---|---|
| Age (years) | - | - | - | - | - |
| Mean ± SD | 50.53 ± 3.55 | 45.67 ± 3.14 | 57.33 ± 2.16 | 32.17 ± 2.32 | 0.002* |
| 95% CI | (49.1-51.9) | (44.4-46.9) | (56.4-58.2) | (30.9-33.5) | - |
| Gender, n (%) | - | - | - | - | - |
| Male | 10 (11.3%) | 12 (13.6%) | 16 (18.1%) | 7 (7.9%) | 0.156 |
| Female | 15 (17.0%) | 13 (14.7%) | 9 (10.2%) | 5 (5.6%) | - |
| Clinical Parameters | - | - | - | - | - |
| PLI Score | 1.953 ± 0.45 | 2.128 ± 0.52 | 3.368 ± 0.68 | 0.511 ± 0.12 | <0.001* |
| GI Score | 2.096 ± 0.48 | 3.116 ± 0.61 | 3.932 ± 0.74 | 0.423 ± 0.09 | <0.001* |
| PPD (mm) | N/A | 5.526 ± 1.25 | 6.434 ± 1.48 | N/A | <0.001* |
Abbreviations: ATH: Atherosclerosis; CP: Chronic Periodontitis; PLI: Plaque Index; GI: Gingival Index; PPD: Probing Pocket Depth; CI: Confidence Interval.
The sex distribution varied across groups, with the ATH group showing a predominance of females (15 females, 10 males), whereas the ATH+CP group demonstrated a predominance of males (16 males, 9 females). The CP group exhibited a relatively balanced sex distribution (13 females, 12 males), and the control group had a slight male majority (7 males, 5 females). Chi-square analysis revealed no statistically significant differences in sex distribution among the groups (p = 0.156).
3.2. Clinical Periodontal Parameters
Comprehensive analysis of clinical periodontal parameters revealed significant differences among the study groups, as detailed in Table 1. The ATH+CP group consistently demonstrated the most severe periodontal conditions across all measured parameters, followed by the CP, ATH, and control groups.
All comparisons between the control group and each disease group revealed statistically significant differences (p < 0.001).
Plaque index scores revealed a transparent gradient of increasing plaque accumulation from the control group (0.511 ± 0.12) to the ATH+CP group (3.368 ± 0.68). Pairwise comparisons revealed statistically significant differences among all groups (p < 0.001), suggesting that atherosclerotic cardiovascular disease may exacerbate plaque accumulation in patients with periodontal disease.
The gingival index scores followed a similar pattern, with the ATH+CP group showing the highest inflammation levels (3.932 ± 0.74), which were significantly greater than those of all the other groups. The CP group (3.116 ± 0.61) presented more severe gingival inflammation than the ATH group (2.096 ± 0.48), whereas the controls maintained a healthy gingival status (0.423 ± 0.09).
Probing pocket depth measurements were applied only to the periodontal disease groups (CP and ATH+CP groups). Compared with the CP group, the ATH+CP group presented significantly deeper pockets (6.434 ± 1.48 mm) (5.526 ± 1.25 mm, p < 0.001), suggesting that concurrent atherosclerotic cardiovascular disease may contribute to more severe periodontal tissue destruction.
3.3. Salivary Lactate Dehydrogenase Levels
The primary outcome analysis revealed highly significant differences in salivary LDH levels among all study groups (F = 95.647,p < 0.001), as illustrated in Fig. (2). The ATH+CP group presented the highest LDH level (24.55 ± 2.55 U/L), followed by the CP group (13.44 ± 4.27 U/L), the ATH group (11.68 ± 3.86 U/L), and the control group (1.53 ± 0.31 U/L) (Table 2).
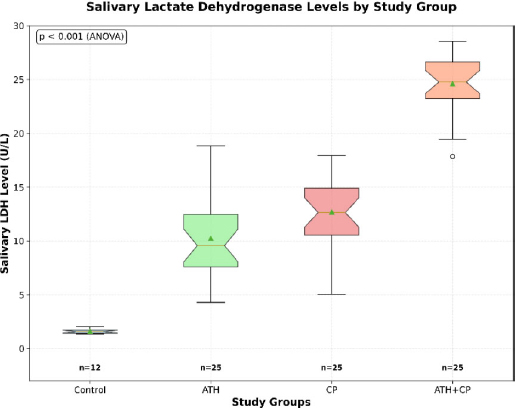
Box plot comparison of salivary lactate dehydrogenase levels among the study groups.
| Group | N | Mean ± SD (U/L) | 95% CI | Median (IQR) | Range | Effect Size vs Control |
|---|---|---|---|---|---|---|
| Control | 12 | 1.53 ± 0.31 | (1.35-1.71) | 1.50 (1.25-1.75) | 1.1-2.2 | - |
| ATH | 25 | 11.68 ± 3.86 | (10.17-13.19) | 11.50 (8.75-14.25) | 5.2-19.8 | 2.89 (Large) |
| CP | 25 | 13.44 ± 4.27 | (11.77-15.11) | 13.20 (10.50-16.80) | 6.1-22.5 | 3.45 (Large) |
| ATH+CP | 25 | 24.55 ± 2.55 | (23.55-25.55) | 24.80 (22.50-26.25) | 19.8-28.5 | 8.95 (Very Large) |
The magnitude of differences between groups was substantial, with effect sizes ranging from large to very large compared with those of the controls. Compared with the control group, the ATH+CP group presented a very large effect size (Cohen's d = 8.95), indicating not only statistical significance but also high clinical relevance.
3.4. Pairwise Group Comparisons
Post hoc analysis via Tukey's HSD test revealed statistically significant differences between all possible group pairs, as detailed in Table 3. The largest difference was observed between the ATH+CP group and the control group (mean difference = 23.02 U/L, p < 0.001), followed by the difference between the CP group and the control group (mean difference = 11.91 U/L, p < 0.001).
| Comparison | Mean Difference (U/L) | 95% CI of Difference | Cohen's d | Effect Size | p-value |
|---|---|---|---|---|---|
| ATH vs CP | 1.76 | (0.85-2.67) | 0.42 | Small | <0.001* |
| ATH vs ATH+CP | 12.87 | (11.45-14.29) | 3.85 | Large | <0.001* |
| ATH vs Control | 10.15 | (8.95-11.35) | 2.89 | Large | <0.001* |
| CP vs ATH+CP | 11.11 | (9.89-12.33) | 3.21 | Large | <0.001* |
| CP vs Control | 11.91 | (10.71-13.11) | 3.45 | Large | <0.001* |
| ATH+CP vs Control | 23.02 | (21.25-24.79) | 8.95 | Very Large | <0.001* |
Notably, even the difference between the ATH and CP groups, which had the smallest absolute difference, remained statistically significant (p < 0.001), indicating that the presence of atherosclerotic CVD alone is associated with measurable increases in salivary LDH levels.
3.5. Gender Analysis
Analysis of sex differences in LDH levels across all participants revealed no statistically significant differences between males (12.85 ± 8.45 U/L) and females (13.49 ± 9.12 U/L, t = 0.602, p = 0.564) (Table 4). This finding was consistent across individual groups, suggesting that sex does not significantly influence salivary LDH levels in either healthy individuals or patients with periodontal disease and/or atherosclerotic cardiovascular disease.
| Gender | N | Mean ± SD (U/L) | 95% CI | T-statistic | p-value |
|---|---|---|---|---|---|
| Male | 45 | 12.85 ± 8.45 | (10.32-15.38) | 0.602 | 0.564 |
| Female | 42 | 13.49 ± 9.12 | (10.67-16.31) |
3.6. Correlation Analysis
Correlation analysis between salivary LDH levels and clinical periodontal parameters revealed varying relationships across the study groups, as presented in Table 5. In the CP group, significant positive correlations were observed between LDH levels and all clinical parameters: PLI (r = 0.103, p = 0.046), GI (r = 0.128, p = 0.041), and PPD (r = 0.240, p = 0.008).
| Parameter | ATH | CP | ATH+CP | Control |
|---|---|---|---|---|
| PLI | - | - | - | - |
| r | 0.291 | 0.103 | 0.059 | -0.077 |
| p-value | 0.252 | 0.046* | 0.006* | 0.746 |
| GI | - | - | - | - |
| r | 0.140 | 0.128 | 0.111 | 0.088 |
| p-value | 0.556 | 0.041* | 0.361 | 0.711 |
| PPD | - | - | - | - |
| r | 0.122 | 0.240 | 0.425 | N/A |
| p-value | 0.609 | 0.008* | 0.002* | N/A |
Abbreviations: ATH: Atherosclerosis; CP: Chronic Periodontitis; PLI: Plaque Index; GI: Gingival Index; PPD: Probing Pocket Depth.
In the ATH+CP group, significant correlations were found between LDH and PLI (r = 0.059, p = 0.006) and between LDH and PPD (r = 0.425, p = 0.002). The strongest correlation was observed between LDH and PPD in the ATH+CP group, suggesting that the severity of tissue destruction is closely related to LDH release in patients with concurrent conditions.
3.7. Diagnostic Accuracy Analysis
Receiver operating characteristic (ROC) analysis was performed to evaluate the diagnostic potential of salivary LDH for the detection of periodontal disease. The analysis demonstrated excellent diagnostic accuracy, with an area under the curve (AUC) of 0.987 (95% CI: 0.965--1.000, p < 0.001), indicating outstanding discriminatory ability, as shown in Fig. (3).
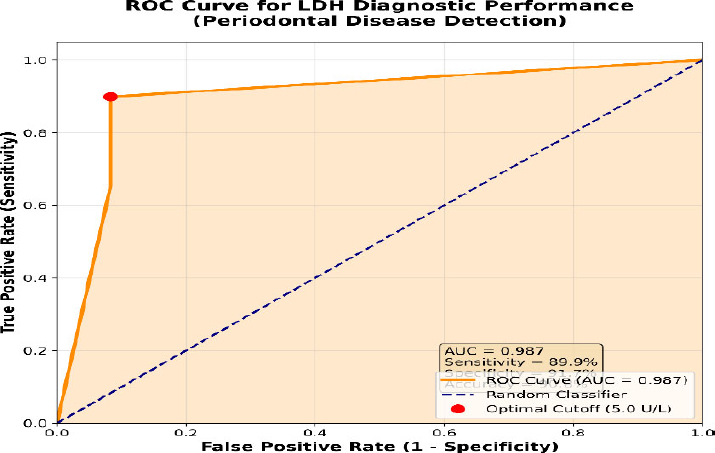
Receiver operating characteristic (ROC) curve for salivary LDH diagnostic performance in periodontal disease detection.
The optimal cutoff value of 5.0 U/L provided the best balance between sensitivity (89.9%) and specificity (91.7%), with an overall accuracy of 90.8% (Table 6). This cutoff demonstrated excellent positive predictive value (98.6%) and moderate negative predictive value (64.7%), making it highly suitable for screening applications.
| Cutoff (U/L) | Sensitivity (%) | Specificity (%) | PPV (%) | NPV (%) | Accuracy (%) | Likelihood Ratio |
|---|---|---|---|---|---|---|
| ≥2.0 | 95.7 | 91.7 | 98.6 | 73.3 | 94.3 | 11.5 |
| ≥5.0 | 89.9 | 91.7 | 98.6 | 64.7 | 90.8 | 10.8 |
| ≥10.0 | 78.3 | 91.7 | 98.6 | 52.4 | 82.8 | 9.4 |
| ≥15.0 | 65.2 | 100.0 | 100.0 | 41.4 | 74.7 | ∞ |
4. DISCUSSION
This research revealed that salivary concentrations of LDH are significantly increased in patients with chronic periodontitis, especially in patients with concomitant chronic periodontitis and atherosclerotic cardiovascular disease. The results of this research support the use of salivary LDH analysis as an additional noninvasive method for assessing periodontal disease and confirming cumulative inflammation in patients with concomitant conditions.
The findings of this research support those of earlier works, which reported increased salivary LDH concentrations in patients with periodontitis [25-28]. The elevated LDH levels confirmed tissue damage in patients with periodontitis. The higher concentrations found in patients with ATH+CP support an additive effect of the disease.
Our results are also supported by those of others in the region. For example, research conducted in Saudi Arabia revealed that there was a significant difference in salivary LDH activity between patients with periodontitis and those without periodontitis, with higher salivary LDH activity in patients with periodontitis [34]. Another study conducted in Iran reported similar results [35].
The positive relationship between salivary LDH and periodontal parameters confirms its role as an indicator of disease severity. The strongest relationship was found between salivary LDH and PPD in patients with ATH+CP, which revealed that with increased severity of periodontitis, there is also an increased value of salivary LDH, especially in patients with concomitant systemic inflammatory diseases such as ATH.
Of note is the high diagnostic accuracy of salivary LDH for periodontal disease, represented by a 0.987 area under the receiver operating characteristic curve. The results indicate that salivary LDH may serve as a potentially useful diagnostic aid in the screening and early detection of periodontal disease, which is imperative to prevent the advancement of disease and its complications [36].
The elevated LDH levels observed in this study can be explained through several interconnected pathophysiological mechanisms. Lactate dehydrogenase, a cytoplasmic enzyme present in all human tissues, serves as a sensitive marker of cellular damage and tissue destruction [37]. In periodontal disease, chronic bacterial infection and subsequent host inflammatory responses lead to extensive tissue damage, resulting in LDH release from damaged periodontal ligament cells, gingival epithelial cells, and inflammatory cells [38, 39].
The inflammatory cascade initiated by periodontal pathogens involves the activation of innate immune responses, including neutrophil recruitment, macrophage activation, and complement system activation [40]. These processes result in the production of inflammatory mediators such as Interleukin-1β (IL-1β), Tumor Necrosis Factor-α (TNF-α), Interleukin-6 (IL-6), and Matrix Metalloproteinases (MMPs), which contribute to tissue destruction and subsequent LDH release [41, 42].
In atherosclerotic cardiovascular disease, similar inflammatory mechanisms contribute to endothelial dysfunction, plaque formation, and myocardial tissue damage [43]. The chronic ischemic conditions associated with coronary artery stenosis led to ongoing myocardial cell damage and death, resulting in continuous LDH release into the systemic circulation and subsequently into the saliva [44]. A correlation between LDH levels and cardiovascular disease severity has been previously established, with elevated levels indicating greater myocardial damage and poorer prognosis [45].
The most striking finding of this study is the synergistic increase in LDH levels in patients with both periodontal disease and atherosclerotic cardiovascular disease. The mean LDH level in the ATH+CP group (24.55 U/L) substantially exceeded what would be expected from simple additive effects of individual conditions, suggesting complex interactions between these disease processes [46].
Several mechanisms may explain this observed elevation. First, periodontal bacteria and their toxins can enter systemic circulation through ulcerated periodontal pockets, contributing to systemic inflammation and accelerating atherosclerotic processes [47, 48]. Bacteremia associated with periodontal disease has been shown to increase inflammatory markers and promote endothelial dysfunction, potentially exacerbating existing cardiovascular disease [49].
Second, shared inflammatory pathways between periodontal and cardiovascular diseases may create a positive feedback loop, where inflammation from one condition amplifies the inflammatory response in the other [50]. This cross-amplification could result in enhanced tissue damage and greater LDH release than would occur with either condition alone [51].
Third, the presence of atherosclerotic cardiovascular disease may compromise the immune system's ability to respond effectively to periodontal infection, leading to more severe periodontal tissue destruction [52]. Conversely, chronic periodontal inflammation may contribute to a systemic inflammatory burden, accelerating atherosclerotic progression and increasing cardiovascular risk [53].
The findings of this study are consistent with those of previous investigations, which have demonstrated elevated LDH levels in patients with periodontal disease [54, 55]. Hamodat and Taha reported similar elevations in LDH levels among patients with chronic periodontitis compared with healthy controls, supporting the utility of this biomarker for periodontal disease assessment [56]. However, their study did not examine patients with concurrent cardiovascular disease, limiting direct comparison with our findings.
Das et al. conducted a comparative evaluation of LDH levels in periodontal diseases and reported significant correlations between LDH levels and disease severity [57]. These results align with our correlation findings, particularly the strong relationship between LDH and probing pocket depth observed in our study groups.
In the context of cardiovascular disease, Chazov et al. evaluated salivary LDH levels in patients with coronary atherosclerosis and reported elevated LDH levels compared with those in healthy individuals [58]. These findings support our observation of increased LDH levels in the ATH group, although their study did not examine patients with concurrent periodontal disease.
The novelty of our study lies in the comprehensive evaluation of patients with both conditions, revealing synergistic effects that have not been previously reported. This finding has important implications for understanding disease interactions and developing integrated treatment approaches [59].
The excellent diagnostic performance of salivary LDH demonstrated in this study has several important clinical implications. The noninvasive nature of saliva collection makes this biomarker particularly attractive for screening applications, especially in populations with limited access to comprehensive periodontal examination [60]. The high sensitivity (89.9%) achieved with the optimal cutoff suggests that salivary LDH could serve as an effective screening tool for identifying individuals who require further periodontal evaluation [61].
Salivary LDH levels showed significant positive correlations with clinical periodontal parameters in the diseased groups, as determined by Pearson's correlation analysis. In the CP group, LDH was significantly correlated with all three clinical parameters, including PLI (r = 0.103, p = 0.046), GI (r = 0.128, p = 0.041), and PPD (r = 0.240, p = 0.008). The most significant correlation of LDH was with the PPD, which directly measures the severity of periodontal tissue destruction and loss of attachment. This is consistent with the mechanistic role of LDH as a cytoplasmic enzyme released during cell death and tissue breakdown [62]. Serial LDH measurements could provide an objective assessment of periodontal healing following therapeutic interventions [63].
For patients with atherosclerotic cardiovascular disease, elevated salivary LDH levels may serve as an early indicator of concurrent periodontal disease, prompting dental referrals and comprehensive periodontal evaluation [64]. This could facilitate early intervention and potentially reduce the risk of cardiovascular complications associated with periodontal disease [65].
The absence of significant sex differences in LDH levels observed in this study is consistent with previous reports and suggests that this biomarker is not influenced by sex-related factors [66]. This finding is important for clinical application, as it indicates that the same diagnostic cutoffs can be applied across both male and female populations without adjustment [67].
The age differences observed among the study groups reflect the natural epidemiology of both periodontal and cardiovascular diseases, which increase in prevalence and severity with increasing age [68]. The older age of the ATH+CP group is consistent with the cumulative nature of both conditions and supports the clinical relevance of our findings in real-world populations [69].
The strong correlations observed between LDH levels and clinical periodontal parameters provide mechanistic insights into the relationship between tissue destruction and biomarker release. The particularly strong correlation with probing pocket depth (r = 0.425, p = 0.002) in the ATH+CP group suggests that LDH release is closely related to the extent of periodontal tissue destruction [70].
Future research should investigate the temporal relationships between LDH elevation and disease progression, potentially through longitudinal studies following patients over extended periods [71]. Additionally, investigations of other inflammatory biomarkers in conjunction with LDH could provide a more comprehensive understanding of the inflammatory networks involved in these disease interactions [72].
The potential for LDH to serve as a treatment monitoring biomarker warrants investigation through intervention studies examining changes in LDH levels following periodontal therapy [73]. Such studies could establish the utility of this biomarker for assessing treatment efficacy and guiding clinical decision-making [74, 75].
5. LIMITATIONS
This study has several limitations that should be acknowledged. First, the cross-sectional design prevents the establishment of causal relationships between salivary LDH levels and disease status. Second, the relatively small sample size, particularly in the control group (n=12), may limit the generalizability of our findings. Third, this was a single-center study conducted in an Iraqi population, which may affect the applicability of the results to other populations. Fourth, despite the study’s comprehensive exclusion criteria, potential residual confounding from unmeasured variables cannot be entirely ruled out. Fifth, the lack of longitudinal follow-up data prevents the assessment of LDH changes over time or in response to treatment. Sixth, multivariate regression analysis was not performed to control for all potential confounders, which should be addressed in future larger studies. Finally, the absence of other cardiovascular biomarkers for comparison limits our ability to assess the relative utility of salivary LDH compared with established markers.
CONCLUSION
Salivary LDH has excellent potential as a noninvasive biomarker for periodontal disease and may reflect the additive inflammatory burden in patients with concurrent atherosclerotic cardiovascular disease.
The substantially elevated LDH levels in patients with both conditions (24.55 ± 2.55 U/L) surpassed the additive effect of each disease alone, providing strong biochemical support for the bidirectional relationship between oral and systemic inflammation.
The diagnostic performance of salivary LDH was outstanding, with an Area Under the ROC curve (AUC) of 0.987. A defined cutoff of 5.0 U/L offers a clinically robust tool, demonstrating high sensitivity (89.9%) and specificity (91.7%) for identifying individuals with periodontal disease. The utility of LDH as an indicator of disease severity was reinforced by its strong positive correlation with clinical parameters, most notably probing pocket depth (r = 0.425, p = 0.002), directly linking biomarker levels to the extent of tissue destruction. Furthermore, the absence of sex-based differences enhances its universal applicability in clinical settings.
These findings have significant implications for integrated healthcare. Salivary LDH testing could serve as a practical screening mechanism in both medical and dental practices. Elevated levels in patients with cardiovascular disease could trigger a dental referral. Moreover, dentists could use this biomarker to assess systemic inflammatory burden and reinforce the importance of oral health in cardiovascular risk management.
Longitudinal research is essential to clarify the temporal relationship between LDH elevation and disease onset. Intervention studies are needed to validate LDH as a marker for monitoring treatment response, and multicenter trials are needed to confirm the generalizability of these findings across diverse populations. In summary, salivary LDH holds significant promise not only for periodontal diagnostics but also as a key biochemical marker highlighting the profound connection between oral health and cardiovascular well-being.
AUTHORS’ CONTRIBUTIONS
The authors confirm contribution to the paper as follows: R.S.H.: Contributed to conceptualization, methodology, investigation, data analysis, writing of the original draft, and writing, review, and editing; M.S.A.: Contributed to investigation, data collection, writing, review, and editing; Z.S.M.: Contributed to data collection, laboratory analysis, writing, review, and editing; S.M.S.: Contributed to supervision, methodology, writing, review, editing, and project administration; S.M.I.: Contributed to supervision, writing, review, and editing. All authors have read and approved the final manuscript.
LIST OF ABBREVIATIONS
| ATH | = Atherosclerotic Cardiovascular Disease |
| AUC | = Area under the curve |
| CAL | = Clinical Attachment Loss |
| CP | = Chronic Periodontitis |
| CRP | = C-reactive Protein |
| ELISA | = Enzyme-linked Immunosorbent Assay |
| GI | = Gingival Index |
| HRP | = Horseradish Peroxidase |
| LDH | = Lactate Dehydrogenase |
| PLI | = Plaque Index |
| PPD | = Probing Pocket Depth |
| ROC | = Receiver Operating Characteristic |
ETHICS APPROVAL AND CONSENT TO PARTICIPATE
This cross-sectional comparative study was conducted between July 2023 and October 2023, following approval from the Ethical Committee of the College of Medicine, University of Kufa (Reference number: MEC-48, Date: 26/6/2023).
HUMAN AND ANIMAL RIGHTS
All human research procedures followed were in accordance with the ethical standards of the committee responsible for human experimentation (institutional and national), and with the Helsinki Declaration of 1975, as revised in 2013.
CONSENT FOR PUBLICATION
After receiving comprehensive information regarding the study objectives, procedures, potential risks, and benefits, all participants provided written informed consent.
AVAILABILITY OF DATA AND MATERIALS
The datasets generated and analyzed during the current study are available from the corresponding author upon reasonable request and are subject to ethical approval and data protection regulations.
ACKNOWLEDGEMENTS
The authors express their gratitude to the participants who volunteered for this study and to the staff at the Al-Sadar Teaching Hospital Heart Center and the Faculty of Dentistry, University of Kufa, for their support in participant recruitment and data collection. We acknowledge the technical assistance provided by the laboratory personnel in conducting the ELISA analyses.


