All published articles of this journal are available on ScienceDirect.
Phytoremediation Powered by Endophytes: Toward a Greener, Sustainable Agriculture
Abstract
Endophytes, microorganisms that live within plant tissues, are emerging as promising, eco-friendly solutions for both environmental remediation and sustainable agriculture. This review examines how these beneficial microbes can be leveraged to address the significant threats of soil and water pollution while reducing the agricultural sector's reliance on synthetic agrochemicals. Endophytes enhance phytoremediation by directly degrading or detoxifying pollutants and by improving the host plant's ability to tolerate stress and absorb contaminants such as heavy metals and pesticides. In agriculture, they boost plant growth by producing phytohormones and enabling biological nitrogen fixation, which in turn reduces the need for chemical fertilizers. They also strengthen plant resilience against biotic and abiotic stressors, such as drought, further diminishing the use of chemical inputs. Despite their considerable potential, challenges remain, including the lack of a centralized database for identification and a limited understanding of host specificity and the dynamics of endophyte communities under different environmental conditions. To fully realize the benefits of endophytes, future research must focus on comprehensive studies that explore their ecological interactions, model their behavior in plant tissues, and address the trade-offs between their positive effects and potential limitations. By integrating endophyte-based strategies, we can couple pollution mitigation with improved crop productivity, aligning with the principles of a circular bioeconomy and advancing sustainable development goals.
1. INTRODUCTION
Phytoremediation uses plants and their associated microorganisms to remove, degrade, or stabilize environmental contaminants and has become an important tool in sustainable environmental management [1]. At its core, this approach is symbiotically enhanced by endophytes, which are microorganisms that live within plant tissues without causing harm [2]. These endophytes play a critical role by promoting plant growth, stress tolerance, and pollutant transformation [3, 4]. Through these functions, endophyte-assisted phytoremediation offers an environmentally friendly approach to mitigating soil and water pollution, key threats to food security and ecosystem stability [5].
Despite increasing interest in this strategy, its broader application in reducing agriculture’s dependence on synthetic agrochemicals remains insufficiently explored. Current research has primarily focused on the direct remediation of heavy metals and organic pollutants [6], leaving a notable gap in our knowledge regarding the full potential of these plant–microbe interactions. This gap limits the integration of phytoremediation into agroecological systems [7].
Recent advances in biotechnology and microbial ecology have revealed the metabolic diversity of endophytes, including their capacity to produce enzymes, bioactive compounds, and signalling molecules involved in pollutant degradation and plant defence [8, 9]. As illustrated in Figure 1, these mechanisms contribute to both environmental remediation and improved plant resilience. These multifaceted benefits underline the potential of integrating endophyte-assisted phytoremediation into broader agricultural and environmental management frameworks. Integrating endophyte-assisted phytoremediation into agricultural management frameworks could therefore link pollution control with enhanced crop productivity and reduced agrochemical inputs. This strategy aligns with the principles of the circular bioeconomy and offers a scalable, cost-effective pathway toward achieving sustainable development goals [10].
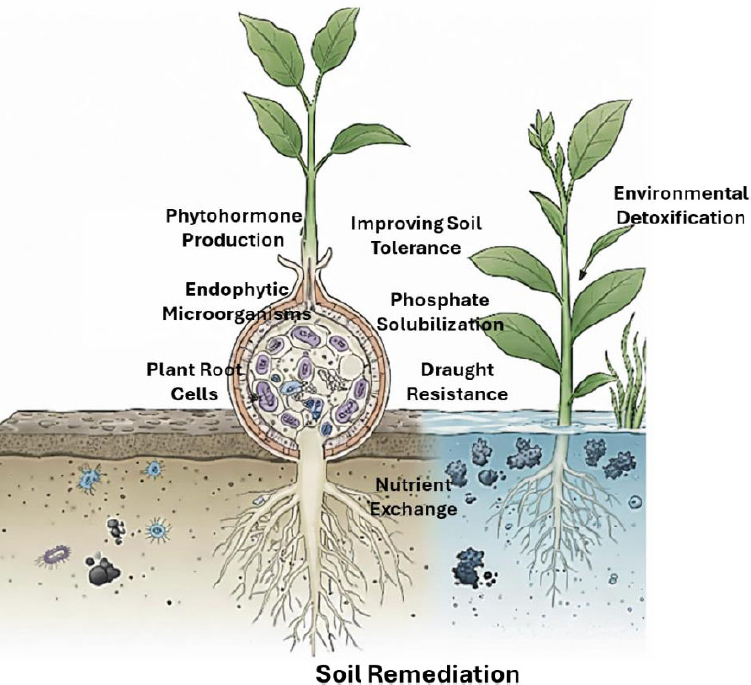
This diagram illustrates the symbiotic relationship where microbial endophytes enhance plant nutrient uptake, nitrogen fixation, and drought resistance. Simultaneously, endophytes facilitate soil remediation by detoxifying organic pollutants and sequestering heavy metals through specialized enzymatic activity. These integrated mechanisms reduce phytotoxicity and bolster environmental health by coupling improved crop productivity with effective pollution mitigation.
2. APPLICATION OF ENDOPHYTES IN AGRICULTURE
Endophyte–host specificity is a complex and context-dependent ecological phenomenon that influences community assembly, functional interactions, and the success of microbial applications in phytoremediation and sustainable agriculture. Host plant identity and genotype have been shown to significantly shape endophyte community composition, with genetic variation in plant hosts correlating with distinct assemblages of root and foliar endophytes, suggesting deterministic host selection processes beyond simple environmental filtering [11, 12]. Conversely, some field surveys indicate that host specificity may be relatively weak in certain ecosystems, with generalist fungal endophytes dominating communities and weak correlations between host phylogeny and endophyte beta diversity, particularly where spatial turnover is low [13]. These contrasting findings highlight a key knowledge gap regarding the mechanisms underlying host specificity, including the roles of plant secondary chemistry, immune signalling, and microhabitat structure. Understanding these mechanisms is crucial because they directly influence how endophytes promote plant growth by facilitating nutrient acquisition, producing phytohormones, alleviating biotic and abiotic stresses, and suppressing phytopathogens [14].
In particular, endophytic bacteria enhance plant development through the synthesis of phytohormones, including auxins, cytokinins, abscisic acid, ethylene, brassinosteroids, and gibberellins, which promote nutrient uptake and biomass accumulation [15]. A study by Hassan [16] demonstrated that both bacterial (Bacillus cereus, Bacillus subtilis) and fungal (Penicillium chrysogenum, Penicillium crustosum) endophytes significantly improved maize (Zea mays L.) growth and yield. In addition, certain endophytic bacteria possess biological nitrogen fixation (BNF) genes that enable them to convert nitrogen gas into other nitrogen forms, such as nitrate and ammonium, which can be utilized by host plants [17]. Nitrogen-fixing endophytes, such as Paenibacillus beijingensis BJ-18, can contribute substantially to host nutrition, supplying 12.9–20.9% of total nitrogen in wheat (Triticum aestivum L.) and 52.2–59.2% in cucumber (Cucumis sativus L.) through biological nitrogen fixation [18].
Beyond direct growth promotion, endophytes significantly contribute to improving soil health and nutrient use efficiency in a way that minimizes environmental impact. For instance, many endophytic fungi and bacteria can solubilize sparingly soluble soil minerals, such as organic and inorganic phosphates, making them bioavailable for the host plant [19]. This microbial action effectively reduces the need for synthetic phosphate fertilizers, mitigating runoff and subsequent eutrophication [20]. Furthermore, certain endophytes produce siderophores, high-affinity iron-chelating compounds, which facilitate iron uptake by the plant in iron-poor soils [21] and concurrently offer a defence mechanism against soilborne pathogens by sequestering essential micronutrients [22]. This multifaceted role in nutrient cycling positions endophytes as indispensable components of sustainable soil management practices.
Beyond nutrient acquisition, endophytic bacteria indirectly enhance plant productivity by improving resistance to biotic and abiotic stresses. Mechanisms include the production of volatile compounds, abscisic acid, ACC deaminase, and indole-3-acetic acid (IAA) [20, 21]. Studies have shown that IAA-producing endophytes such as Bacillus subtilis, Enterobacter ludwigii, Klebsiella sp., and Pseudomonas fluorescens significantly increase crop yield and stress tolerance across multiple species.
Beyond nutrient acquisition, endophytic bacteria indirectly enhance plant productivity by improving resistance to biotic and abiotic stresses [23]. Mechanisms include the production of volatile compounds, abscisic acid, ACC deaminase, and indole-3-acetic acid (IAA). Studies have shown that IAA-producing endophytes such as Bacillus subtilis, Enterobacter ludwigii, Klebsiella sp., and Pseudomonas fluorescens significantly increase crop yield and stress tolerance across multiple species [14, 16, 15, 17]. A study by Lally et al. [24] showed that individual Pseudomonas fluorescens endophytic strains significantly improved the growth of rapeseed (Brassica napus) under greenhouse and field conditions [24]. Therefore, endophytes effectively improve plant growth and tolerance to biotic and abiotic stress, thus reducing agrochemicals and supporting sustainable agricultural practices without harming soil quality.
3. ENVIRONMENTAL RELEVANCE
Endophytes are known for their role in plant well-being through improving the metabolic capacity of plants [25]. Moreover, endophytes regulate the absorption of contaminants, such as metals and pesticides, through various mechanisms, including biosorption, sequestration, and transformation [6, 26]. It was found that endophytes enhance the bioaccumulation capacity of metals [27], whereas it was emphasized that endophytes have the potential to degrade pesticides. Nevertheless, their efficiency in regulating contaminants may require some fortification, which can affect metabolic pathways [28].
The detoxification function of endophytes is highly dependent on specialized enzymatic activity that enables the breakdown of persistent organic pollutants (POPs) within the plant–microbe association. These microorganisms produce oxidoreductase enzymes, particularly laccases, peroxidases, and dioxygenases, that catalyze the degradation and mineralization of persistent organic pollutants, including polycyclic aromatic hydrocarbons and recalcitrant pesticides [29, 30]. This mechanism is a critical element of phytotransformation, allowing the host plant to either absorb less harmful modified substances or achieve full breakdown of the compounds within plant tissues. Furthermore, endophytes enhance the host plant's overall stress resilience, protecting against both abiotic factors (e.g., heavy metals) and biotic threats by initiating defence mechanisms like systemic acquired resistance (SAR) [31]. This synergistic effect significantly bolsters the plant's capacity to survive and execute effective remediation in highly polluted environments, a capability frequently demonstrated by strains isolated directly from hyperaccumulator plants [32].
On the other hand, endophytes have been found to mitigate the effects of drought and salinity in plants [33, 34], with different organs, such as the rhizosphere and leaves, exhibiting varying responses. The endophytes help in the synthesis of vital compounds and nutrient acquisition [35], hence fortifying plants against environmental stress, including climate change. Despite the potential of endophytes to enhance plant resilience to environmental stressors, their capacity may be compromised at extreme levels [36]. It was reported that there is a decrease in endophyte diversity with increasing metal concentration [37]. Moreover, there is a decrease in fungal endophytes and an increase in bacterial endophytes after exposure to extreme metal concentrations [38]. The defence mechanisms of the hosts were found to be enhanced at the expense of reproductive potential [39]. Despite their demonstrated benefits, systematic studies evaluating trade-offs between enhanced stress tolerance, growth, and reproductive fitness remain limited. Moreover, there is still a need to model their progression within plant tissues to provide an understanding of their behaviour and responses to specific stressors in this Anthropocene era.
Building upon the specific environmental applications discussed, the mechanisms employed by endophytes-from pollutant detoxification to stress resilience-confirm their significant potential. These cumulative findings are synthesized in Table 1, which highlights the dual roles of endophytes in enhancing crop productivity and supporting ecosystem remediation. Recent studies provide strong evidence that endophytes confer measurable growth and stress mitigation benefits to crops under both controlled and field conditions. For example, rice plants inoculated with the endophytic bacterium Pseudomonas mosselii PR5 consistently showed significantly increased growth, nutrient acquisition (including N, P, Mg, and Zn), and grain yield compared with uninoculated controls across multiple application methods, including seedling priming and root drenching [40]. These findings demonstrate the potential of beneficial endophytes to improve crop productivity while enhancing nutrient uptake in agriculturally relevant systems.
| Endophyte Function Category | Specific Mechanism/Compound | Role in Sustainable Agriculture (Growth Promotion & Health) | Role in Environmental Remediation (Phytoremediation) |
|---|---|---|---|
| Nutrient Management | Biological Nitrogen Fixation (BNF) | Converts nitrogen gas to other nitrogen forms such as nitrate and ammonium, reducing the need for chemical fertilizers1. For example, Paenibacillus beijingensis BJ-18 provided up to 20.9% of the total nitrogen to wheat. | N/A |
| - | Mineral Solubilization | Solubilizes sparingly soluble soil minerals, such as organic and inorganic phosphates, making them bioavailable to the host plant. This action mitigates runoff and subsequent eutrophication. | N/A |
| - | Siderophores | Produces high-affinity iron-chelating compounds that facilitate iron uptake by the plant in iron-poor soils. | Concurrently offers a defence mechanism against soilborne pathogens by sequestering essential micronutrients. |
| Plant Stress Resistance | Phytohormone Production | Produces phytohormones (e.g., auxins, cytokinins, abscisic acid, ethylene, brassinosteroids, and gibberellins), which promote growth and nutrient absorption. | Enhances the host plant's overall stress resilience against abiotic factors, such as heavy metals. |
| - | Drought Mitigation | Supports plants against drought via the production of volatile compounds, abscisic acid, and the enzyme ACC deaminase. | N/A |
| - | Systemic Defence | Indirectly promotes plant productivity by enhancing plant resilience against biotic and abiotic stresses. | Bolsters defence by initiating mechanisms like Systemic Acquired Resistance (SAR) against biotic threats. |
| Pollutant Remediation | Detoxifying Enzymes | N/A | Synthesizes specialized oxidoreductase enzymes (specifically laccases, peroxidases, and dioxygenases) that catalyse the initial degradation and ultimate mineralization of hazardous organic compounds. |
| - | Contaminant Mechanisms | N/A | Regulates the absorption of contaminants (e.g., metals and pesticides) through mechanisms such as biosorption, sequestration, and transformation. |
| - | Pollutant Examples | N/A | Degrades substances such as polycyclic aromatic hydrocarbons (PAHs) and certain stubborn pesticides. Examples include Serendipita indica for arsenic removal and the bacterial strain Rhodococcus sp. BCH2 for acetamiprid degradation. |
Endophytic fungi can also play dual roles as growth promoters and biocontrol agents. In wheat (Triticum aestivum), the fungal endophyte Epicoccum layuense J4-3 significantly inhibited the mycelial growth of the pathogen Fusarium graminearum and reduced disease severity under both greenhouse and field conditions; importantly, plants treated with this endophyte exhibited enhanced growth metrics such as shoot length, root length, and biomass relative to untreated controls [41]. This example illustrates how endophytes can simultaneously improve plant health and suppress key pathogens, contributing to yield stability in cereal crops.
Additional research on seed endophytes has shown that bacterial communities associated with barley seeds (e.g., Bacillus subtilis, B. licheniformis, and B. pumilus) can promote early seedling growth by increasing root and shoot length and may also inhibit pathogens such as Fusarium graminearum in vitro, supporting their potential use in dual growth promotion and disease management strategies [42]. Although much of this work remains at the early growth stage or in vitro, such findings underscore the functional diversity of endophytic bacteria and their capacity to contribute to both plant growth and defence.
Mechanistically, these beneficial effects are often linked to endophyte-mediated nutrient cycling, phytohormone production, and antagonism against pathogens, reflecting their multifaceted roles in supporting plant performance under diverse environmental conditions. Despite the growing enthusiasm for endophyte-based solutions in sustainable agriculture and phytoremediation, their deployment raises ecological and regulatory concerns that must be critically addressed. Introduction of non-native or engineered microorganisms can disrupt native soil microbial communities, potentially altering biodiversity, nutrient cycling, and ecosystem functioning in unpredictable ways [43, 44]. For example, shifts in microbial community composition following microbial inoculation have been documented and may not revert to pre-treatment states, suggesting lasting impacts on soil ecology [43].
From a regulatory standpoint, standard frameworks in many regions (e.g., plant protection and fertiliser laws) were not initially designed for living microbial products, leading to unclear approval pathways and inconsistent biosafety evaluations for microbial inoculants, including endophytes [45, 46]. Moreover, the potential for horizontal gene transfer among introduced microbes and native populations raises concerns about the spread of undesirable genetic traits, such as antibiotic resistance, particularly if safety assessments are incomplete [47]. These biological and regulatory uncertainties underscore the need for rigorous environmental risk assessments, clear safety standards (e.g., identification, toxicity, and non-target impact testing), and harmonised regulatory frameworks to ensure that endophyte applications deliver ecological benefits without compromising soil health, biodiversity, or long-term ecosystem stability.
Taken together, these contemporary examples provide quantitative evidence that endophyte-mediated strategies can enhance crop growth, nutrient use efficiency, stress tolerance, and resilience-key attributes for integrating microbial solutions into sustainable agricultural and phytoremediation practices.
4. CHALLENGES AND LIMITATIONS
Although research on endophytes has advanced substantially, the absence of an integrated database for reliable identification remains a major limitation, particularly when dealing with highly diverse communities with bioremediation potential [48]. A large proportion of endophyte species remain unidentified, reflecting the immense and largely unexplored diversity of these microorganisms. This taxonomic uncertainty complicates the development of predictive models describing endophyte function across plant species and developmental stages. Although host specificity is fundamental in ecology, it remains poorly understood in endophyte–plant associations, where factors such as climate, temperature, and host physiology strongly influence endophyte abundance [49]. As a result, it may be difficult to employ similar techniques across different ecological regions.
Organ traits and types, host species, and seasonality have also been found to influence the abundance of endophytes [50]. Overcoming current limitations in endophyte identification, host-specificity modelling, and mechanistic insight requires the integration of high-throughput omics technologies. Metagenomics, in particular, bypasses cultivation constraints by capturing the full genetic diversity of endophyte communities directly within plant tissues [51]. Complementary use of transcriptomics and metabolomics provides the dynamic and functional data necessary for predictive modelling. Transcriptomics reveals the specific host and microbial genes being expressed, while metabolomics identifies and quantifies the bioactive compounds (metabolites) produced during stress mitigation and growth promotion [52, 53]. By integrating these molecular datasets, researchers can move beyond single-strain application toward developing effective, tailored multi-endophyte consortia that are robust under dynamic field conditions, offering precision agriculture tools necessary to fully leverage endophyte technology.
Advancements in omics technologies, including metagenomics, transcriptomics, and metabolomics, have greatly enhanced our capacity to elucidate the complex molecular and ecological mechanisms underpinning endophyte-assisted phytoremediation and plant productivity (Fig. 2). Metagenomics enables high-resolution profiling of total microbial communities within plant tissues, revealing endophytic diversity and functional gene pools that would otherwise remain inaccessible due to cultivation limitations [54]. For example, metagenomic sequencing has uncovered extensive taxonomic and functional diversity of bacterial and fungal endophytes and identified genes involved in nutrient cycling, stress response, and secondary metabolite synthesis that may contribute to phytoremediation efficacy and plant resilience [44].
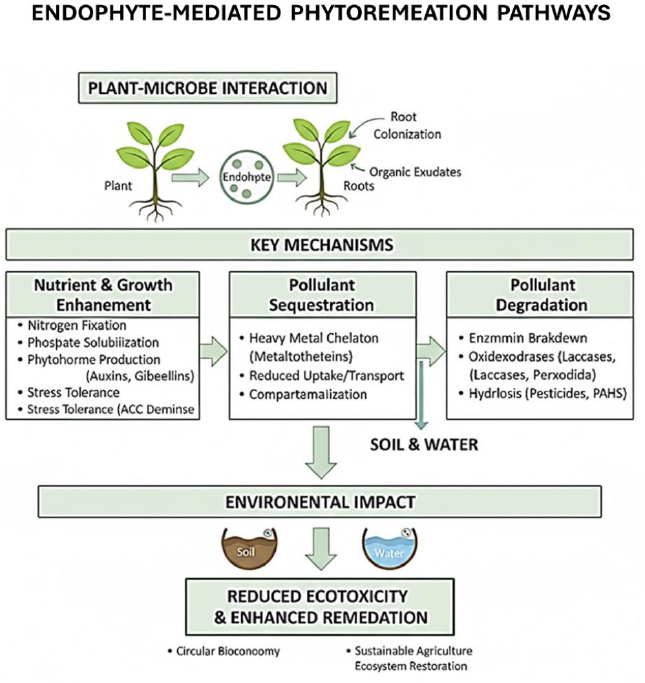
Conceptual framework of endophyte-mediated phytoremediation pathways for sustainable environmental management and circular bioeconomy.
Despite its power, metagenomic analysis faces limitations. The overwhelming presence of host plant DNA in tissue samples can mask microbial sequences, making assembly and annotation challenging without optimized enrichment steps [54]. Additionally, incomplete reference databases for many microbial groups, especially fungal endophytes, restrict confident functional assignment, and rare but ecologically important taxa may be underrepresented in assembled metagenomes [44].
Transcriptomics (and metatranscriptomics) complements metagenomics by capturing the active gene expression profiles of both host plants and associated microbes under specific environmental conditions or pollutant exposures. Transcriptomic data help distinguish between mere genetic potential and real-time functional activity, shedding light on stress-responsive genes, detoxification pathways, and symbiotic signalling networks that are engaged during phytoremediation or abiotic stress mitigation. Although direct plant–endophyte metatranscriptomic datasets remain limited, scoping analyses indicate that transcriptome profiling is a promising tool for identifying key molecular responses in plant–microbe interactions [55]. A major limitation of transcriptomics is the transient nature of RNA, which requires stringent sampling and preservation protocols to avoid degradation and ensure reliable expression data.
Metabolomics adds another functional layer by identifying and quantifying small molecules produced by plant–microbe interactions, including phytohormones, antioxidants, chelators, and other bioactive compounds that mediate stress tolerance and pollutant transformation. Studies comparing root endophyte compositions and associated metabolite profiles in different plant genotypes illustrate how metabolomics can link microbial activity to phenotypic outcomes such as disease resistance or stress tolerance [56]. However, the chemical diversity and dynamic range of metabolites present analytical challenges; no single detection platform captures the entire metabolome, requiring a combination of mass spectrometry and nuclear magnetic resonance techniques that are resource-intensive and generate complex data.
The integration of multi-omics datasets linking metagenomic potential, transcriptomic activity, and metabolomic outcomes holds the greatest promise for advancing predictive models of endophyte function in phytoremediation and sustainable agriculture. Such holistic frameworks can identify candidate microbial consortia with synergistic gene expression profiles and metabolite signatures tailored to specific soil pollutants or crop stress conditions [57, 58]. Nonetheless, multi-omics integration remains computationally demanding and requires robust bioinformatics pipelines capable of reconciling different data types, temporal scales, and environmental variables. Standardization of experimental designs, metadata reporting, and analytical workflows is urgently needed to ensure reproducibility and comparability across studies.
In summary, omics approaches offer unprecedented insights into the structure, function, and dynamics of endophyte–plant systems relevant to phytoremediation. Yet, technical limitations such as host DNA interference, transient RNA stability, metabolite detection bias, and data integration complexity pose ongoing challenges. Overcoming these hurdles through optimized sampling strategies, expanded reference databases, and advanced analytical tools will be essential to fully harness omics technologies for precision endophyte applications in sustainable agricultural and environmental management.
A study reported high endophyte abundance in the stem compared to the leaves and roots, with diversity also varying across organs [59]. However, information on the interactive effect of the aforementioned parameters on endophytes and factors influencing inter-tissue transfer is still lacking. Despite distribution across organs and host species, with seasonal variation, [60] indicated that endophytes may change at different plant growth stages due to the different roles they play at each stage, whereas [49] reported an inconclusive trend for host specificity. The roles of host life cycle and phylogeny in shaping the spatial distribution of endophytes remain unclear. Therefore, there is a need for a comprehensive study to understand the evolutionary and ecological pressures that drive the dynamics of endophytes and their transport mechanisms.
Despite endophytes showing the capacity to enhance host plant resilience to environmental stressors, there are some limitations. Mai et al. [61] reported that endophyte effectiveness may vary seasonally due to climate-driven ecological dynamics, whereas [62] indicated that the selection of endophyte strains with good capacity for bioaccumulation and chelation may be a tedious exercise, as most remain unidentified. Moreover, regulatory frameworks governing endophyte application remain underdeveloped, despite growing evidence of their potential for alleviating plant stress [63]. Nevertheless, more attention should be paid to their toxicant biotransformation capacity to ensure that the complex compounds formed do not result in cumulative effects over time and become a future problem.
CONCLUSION AND FUTURE PERSPECTIVES
The integration of microbial endophytes offers a novel and sustainable strategy for simultaneously enhancing environmental remediation and agricultural productivity. Endophytes contribute uniquely by promoting plant growth, facilitating nutrient acquisition, mitigating biotic and abiotic stresses, and supporting pollutant detoxification, positioning them as eco-friendly alternatives to synthetic agrochemicals. Their application also represents a paradigm shift in phytoremediation, as they enable plants to tolerate and transform contaminants while maintaining ecosystem functionality. However, this approach is challenged by ecological complexities, including reduced endophyte diversity under extreme metal stress and shifts in host defence allocation, which can limit functional outcomes.
To fully harness their potential, future research should prioritize the design of multi-endophyte consortia capable of functioning across diverse environmental conditions, coupled with rigorous long-term field validation to ensure stability, efficacy, and ecological safety. Mechanistic studies are also needed to address trade-offs, host specificity, and the biotransformation of potentially harmful compounds. On the technological and regulatory front, establishing proactive frameworks for monitoring inoculant safety, biosafety, and environmental impacts is essential. By advancing these research, technological, and policy directions, microbial endophytes can be established as a cornerstone of the circular bioeconomy, linking pollutant mitigation with improved crop productivity and fostering a truly sustainable agricultural future.
AUTHORS’ CONTRIBUTIONS
The authors confirm contribution to the paper as follows: M.C.M., J.L., and N.M.D.B.: Conceptualization, investigation, writing the original draft, review, editing, and approval of the final version. All authors have conceptualized the study, collected the data, and written the original manuscript. Finally, authors have edited, reviewed, and approved the final manuscript.
LIST OF ABBREVIATIONS
| BNF | = Biological Nitrogen Fixation |
| POPs | = Persistent Organic Pollutants |
| SAR | = Systemic Acquired Resistance |
| PAHs | = Polycyclic Aromatic Hydrocarbons |
ACKNOWLEDGEMENTS
All authors express their sincere appreciation to Sefako Makgatho University.


